Blood Pressure Watch Guide: How PPG Wearables Measure BP and What to Know Before You Buy
A blood pressure watch uses PPG sensors to estimate BP from your wrist. Learn how they work, which models are available, accuracy limits, and buying tips.
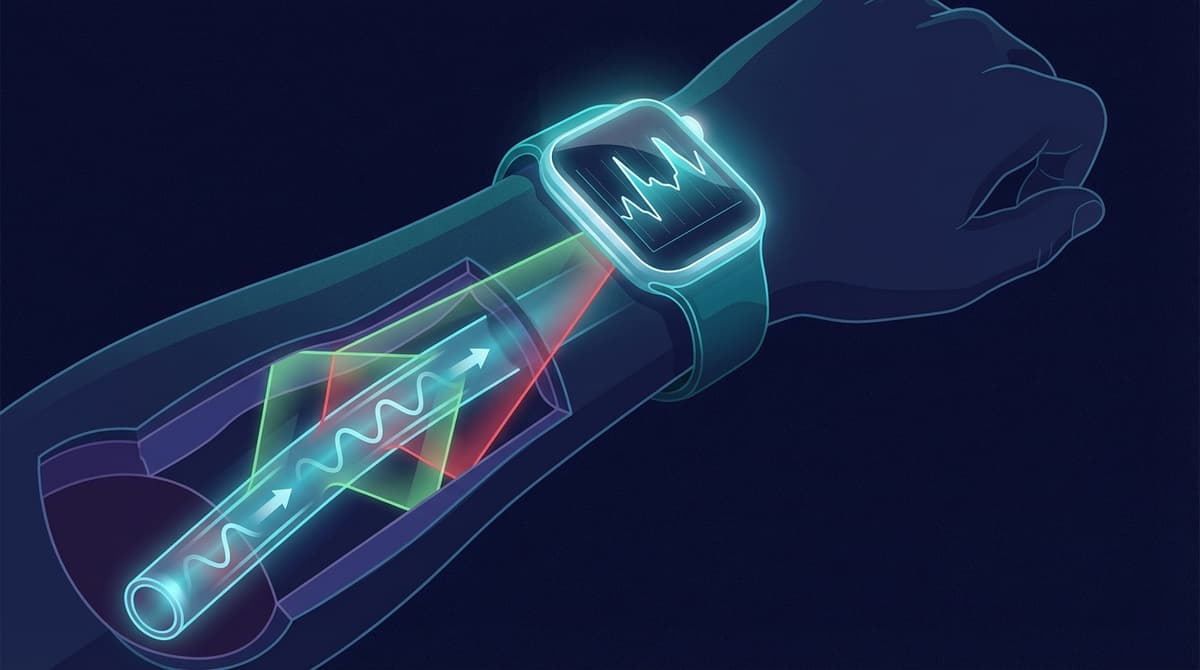
A blood pressure watch is a wrist-worn wearable that uses optical PPG sensors to estimate your blood pressure without a traditional inflatable cuff. These devices work by analyzing changes in the light reflected from blood vessels in your wrist to infer arterial pressure. While the technology is advancing quickly, current blood pressure watches are best understood as trend-tracking tools rather than clinical-grade monitors.
How Does a Blood Pressure Watch Work?
Every blood pressure watch on the market today relies on photoplethysmography, or PPG. A small LED on the underside of the watch shines light into the skin, and a photodetector measures how much light is absorbed or reflected by the pulsating blood in your capillaries and arterioles. The resulting PPG signal is a waveform that rises and falls with each heartbeat.
The shape of that waveform carries information about the state of your cardiovascular system. Features like the steepness of the systolic upstroke, the position of the dicrotic notch, and the rate of diastolic decay all reflect arterial stiffness, vascular resistance, and the pressure driving blood through the arteries. For a detailed look at how these waveform features are identified, see our guide on PPG morphology features.
Blood pressure watches use one of two main approaches (or a combination of both) to translate the PPG signal into a blood pressure estimate.
Pulse Wave Analysis (PWA)
Pulse wave analysis examines the shape of each PPG pulse at a single measurement site, typically the wrist. Algorithms extract dozens of features from the waveform contour and feed them into a mathematical model that outputs systolic and diastolic blood pressure estimates. The Samsung Galaxy Watch series uses this approach.
The underlying principle is that arterial stiffness affects both blood pressure and the PPG waveform shape. When blood pressure rises, arteries stiffen, and the pulse wave travels faster and reflects differently. These changes show up as subtle shifts in the PPG waveform. A thorough explanation of the signal characteristics involved is available in our PPG waveform basics article.
Elgendi et al. (2019) reviewed the use of PPG for hypertension assessment and identified over 30 waveform features with statistically significant correlations to systolic and diastolic blood pressure, though the strength of these correlations varies widely across individuals (DOI: 10.1038/s41746-019-0136-7).
Pulse Transit Time (PTT)
Pulse transit time measures how long the pressure wave takes to travel between two points in your body. Because stiffer arteries (associated with higher blood pressure) conduct the pulse wave faster, shorter transit times generally indicate higher pressure.
Some blood pressure watches combine PPG with other sensors to estimate PTT. For example, a watch might use its PPG sensor alongside an electrical biosensor to capture ECG-like timing. The interval from the electrical heart signal to the arrival of the pulse at the wrist provides a proxy for PTT.
Mukkamala et al. (2015) showed that PTT-based methods typically achieve systolic blood pressure errors of 8 to 12 mmHg (standard deviation), which falls outside the accuracy requirements set by the AAMI/ANSI SP10 standard of 5 plus or minus 8 mmHg (DOI: 10.1109/TBME.2015.2441951). Our in-depth article on PPG blood pressure estimation methods covers the technical details of PTT and PWA approaches.
Which Watches Can Measure Blood Pressure?
The market for blood pressure watches is still relatively small compared to heart rate or SpO2-capable wearables. Here are the most notable devices as of early 2026.
Samsung Galaxy Watch Series
Samsung has been the most aggressive major manufacturer in this space. The Galaxy Watch 7, Watch Ultra, and newer models include a blood pressure monitoring feature that uses PPG-based pulse wave analysis. The feature was first introduced in South Korea with regulatory approval from MFDS (the Korean equivalent of the FDA) and has since expanded to additional markets.
Samsung's approach requires users to calibrate the watch against a standard cuff-based monitor every two to four weeks. After calibration, the watch can take on-demand BP readings. Samsung reports that the device meets the accuracy threshold set by Korean medical device standards, though independent validation data across diverse populations remains limited.
Huawei Watch D Series
Huawei took a different approach with the Watch D and Watch D2, integrating a miniaturized inflatable cuff into the watch band itself. This is technically an oscillometric measurement rather than a pure PPG approach, and it has received medical device certification in China and parts of Europe. The result is closer to a traditional cuff measurement but with the trade-off of a bulkier band and a measurement experience that involves inflation.
Other Entrants
Several smaller companies and startups have released or announced blood pressure watches using various PPG-based methods. These include devices from companies like Aktiia (wrist-worn, calibration-required, CE-marked in Europe) and Biobeat (patch and wristwatch formats with CE and FDA clearance for specific clinical indications). The space is evolving rapidly, and new devices continue to enter the market.
How Accurate Are Blood Pressure Watches?
This is the most important question for anyone considering a blood pressure watch, and the honest answer is: not accurate enough for clinical decision-making in most cases.
The international standard for non-invasive blood pressure devices (ISO 81060-2) requires a mean error of no more than 5 mmHg with a standard deviation of no more than 8 mmHg, validated across a population that includes a range of blood pressure levels, ages, and arm sizes. Very few wrist-worn PPG-based devices have been validated to this standard using independent protocols.
Stergiou et al. (2022) published a position statement from the European Society of Hypertension reviewing cuffless blood pressure devices. The authors concluded that while the technology shows promise, most cuffless devices have not been validated according to established protocols, and the evidence supporting their clinical use is insufficient (DOI: 10.1097/HJH.0000000000003224). They specifically noted that calibration-dependent devices may appear accurate shortly after calibration but drift over days to weeks.
Why Wrist Measurement Is Harder
The wrist is a particularly challenging site for blood pressure estimation. The radial and ulnar arteries at the wrist are smaller than the brachial artery in the upper arm, and the PPG signal from the wrist is more susceptible to motion artifacts, skin tone variation, and differences in how tightly the watch sits. Hydration, ambient temperature, and even caffeine intake can alter peripheral vascular tone and change the PPG waveform in ways that mimic or mask blood pressure changes.
For a broader discussion of how consumer wearables compare to clinical devices, see our article on clinical-grade vs. consumer wearables.
The Calibration Problem
Most blood pressure watches require periodic calibration against a traditional cuff device. This creates an inherent limitation: the watch is only as good as its ability to track changes from the last calibration point. If your baseline blood pressure shifts (due to medication changes, stress, illness, or simply time), the watch readings will drift until you recalibrate.
Some newer machine-learning approaches aim to reduce or eliminate the calibration requirement, but calibration-free accuracy remains an unsolved problem. The relationship between the PPG waveform and blood pressure is subject-specific, meaning a model trained on population-level data will always have a margin of error for any given individual. Our article on continuous blood pressure monitoring from PPG explores this challenge in more detail.
What Is the FDA and Regulatory Status?
Regulatory clearance for blood pressure watches varies significantly by country. Understanding the regulatory picture is important because it signals how much scrutiny a device's accuracy claims have received.
United States (FDA)
As of early 2026, no wrist-worn blood pressure watch has received full FDA 510(k) clearance as a stand-alone blood pressure measurement device. The FDA classifies blood pressure devices under a well-established regulatory pathway with specific accuracy requirements. Some wearable devices have received FDA clearance through the De Novo pathway for blood pressure "trending" or "monitoring" rather than absolute measurement, an important distinction.
In September 2024, the FDA issued a safety communication warning consumers that smartwatches and rings claiming to measure blood pressure without calibration have not been reviewed or authorized by the FDA and may provide inaccurate results.
South Korea (MFDS)
South Korea has been more open to clearing PPG-based blood pressure features. Samsung's blood pressure monitoring app received MFDS clearance, enabling the feature for Korean users and serving as a stepping stone for expansion into other markets.
Europe (CE Marking)
Several devices, including the Aktiia bracelet and Biobeat system, have received CE marking under the European Medical Device Regulation. CE marking requires clinical evidence of safety and performance, though the specific accuracy thresholds and validation protocols can differ from those required by the FDA.
Practical Buying Advice for Blood Pressure Watches
If you are considering purchasing a blood pressure watch, here are the key factors to evaluate.
Check for Published Validation Data
The single most important thing you can do is look for independent validation studies. A device that has been tested by researchers outside the company, using a standardized protocol with a reasonable sample size (at least 85 subjects per ISO 81060-2), is far more trustworthy than one backed only by marketing claims. Look for published papers in peer-reviewed journals, not just press releases.
Understand What the Device Actually Measures
There is a significant difference between a device that claims to give you a blood pressure number (e.g., "120/80 mmHg") and one that tracks relative changes or trends. A trend-tracking device might tell you that your blood pressure is higher today than yesterday, which can be useful, but it cannot reliably tell you whether you are hypertensive.
Know the Calibration Requirements
Ask how often the device needs calibration, what type of reference device is required, and how much accuracy degrades between calibrations. If you do not own a cuff-based monitor for calibration, you will need to buy one anyway, which somewhat defeats the purpose of a cuffless device.
Consider Your Use Case
Blood pressure watches are most useful for:
- Trend monitoring: Tracking how your blood pressure responds to exercise, diet changes, or medication over time
- Convenience screening: Getting a rough sense of your BP level between doctor visits
- Data collection: Logging frequent readings to share with your healthcare provider
They are not appropriate for:
- Diagnosing hypertension: A clinical evaluation with a validated device is required
- Medication titration: Adjusting medications based on watch readings alone is risky
- Emergency assessment: Do not rely on a blood pressure watch to detect a hypertensive crisis
Expect the Technology to Improve
The field of cuffless blood pressure measurement is one of the most active areas in wearable health technology. Machine learning models are getting more sophisticated, sensor hardware is improving, and regulatory pathways are becoming clearer. A blood pressure watch you buy today may be significantly outperformed by models released in the next two to three years. Think of early-generation devices as useful but imperfect tools.
What the Research Says About the Future
PPG-based blood pressure estimation is the subject of hundreds of active research programs. Several directions look promising.
Deep learning models trained on large, diverse datasets are beginning to reduce the gap between PPG-based estimates and cuff-based measurements. Transfer learning techniques allow models to be partially personalized with a small number of calibration readings, rather than requiring extensive per-user training data.
Multi-modal sensing, combining PPG with accelerometer data, bioimpedance, skin temperature, and electrodermal activity, provides additional physiological context that can improve blood pressure estimates. Some next-generation devices are integrating these additional sensors alongside PPG.
However, the fundamental challenge remains: the PPG signal at the wrist is an indirect proxy for central blood pressure, and the relationship between the two depends on dozens of physiological variables that change over time. As Mukkamala and colleagues noted in their 2015 review, the path to clinical-grade cuffless BP measurement requires not just better algorithms but better understanding of the underlying physiology.
Frequently Asked Questions
How accurate are blood pressure watches compared to a traditional cuff?
Most blood pressure watches show higher error margins than validated arm cuffs. Independent studies typically report mean absolute errors of 8 to 12 mmHg for systolic pressure, which is above the AAMI/ANSI threshold of 5 plus or minus 8 mmHg. Accuracy is generally better right after calibration and degrades over time.
Does the Samsung Galaxy Watch measure blood pressure?
Yes. The Samsung Galaxy Watch (Ultra, Watch 7, and newer models) includes blood pressure monitoring via PPG-based pulse wave analysis. The feature requires calibration with a traditional cuff every two to four weeks and is available in South Korea, with limited availability in other regions.
Can a blood pressure watch replace my doctor's cuff monitor?
No. Blood pressure watches can be helpful for tracking trends and patterns between clinic visits, but they should not be used as a substitute for validated clinical devices when diagnosing or managing hypertension. Always discuss your readings with your healthcare provider.
Do blood pressure watches need calibration?
Most current blood pressure watches require periodic calibration against a traditional cuff device. Calibration intervals vary by manufacturer but are typically every two to four weeks. Some devices require calibration only at initial setup, while others prompt for recalibration more frequently.
What is pulse wave analysis in a blood pressure watch?
Pulse wave analysis is a technique that examines the detailed shape of each PPG pulse captured at your wrist. The algorithm extracts features such as the steepness of the systolic upstroke, the timing and depth of the dicrotic notch, and the overall pulse contour. These features correlate with arterial stiffness and blood pressure, allowing the watch to estimate your BP from optical data alone.
Are any blood pressure watches FDA-cleared?
As of early 2026, no wrist-worn smartwatch has received full FDA clearance for standalone cuffless blood pressure measurement. Some wearable devices have received limited clearance for blood pressure trending or monitoring. The FDA has warned consumers about devices making unauthorized blood pressure claims.
What should I look for when buying a blood pressure watch?
Prioritize devices with published, independent validation studies in peer-reviewed journals. Check the regulatory status in your country, understand the calibration requirements, and be clear about whether the device provides absolute blood pressure numbers or relative trend data. A device backed by clinical evidence is worth more than one with impressive marketing.
Frequently Asked Questions
- How accurate are blood pressure watches compared to a traditional cuff?
- Most blood pressure watches show higher error margins than validated arm cuffs. Studies report mean errors of 8 to 12 mmHg for systolic pressure, which exceeds the AAMI/ANSI threshold of 5 plus or minus 8 mmHg required for clinical-grade devices.
- Does the Samsung Galaxy Watch measure blood pressure?
- Yes. The Samsung Galaxy Watch (Ultra, Watch 6 and 7 series) includes a blood pressure monitoring feature using PPG-based pulse wave analysis, but it requires periodic calibration with a traditional cuff and is currently cleared in South Korea and select markets.
- Can a blood pressure watch replace my doctor's cuff monitor?
- No. Blood pressure watches are useful for tracking relative trends over time but should not replace validated clinical devices for diagnosing or managing hypertension.
- Do blood pressure watches need calibration?
- Most do. Calibration against a standard cuff device is typically required at initial setup and periodically afterward, often every two to four weeks, to maintain reasonable accuracy.
- What is pulse wave analysis in a blood pressure watch?
- Pulse wave analysis examines the shape of the PPG waveform captured at your wrist, extracting features like the systolic rise time, dicrotic notch position, and pulse contour that correlate with arterial stiffness and blood pressure.
- Are any blood pressure watches FDA-cleared?
- As of early 2026, no wrist-worn blood pressure watch has received full FDA clearance for standalone cuffless blood pressure measurement. Some devices have De Novo or 510(k) clearance for blood pressure trending rather than absolute measurement.
- What should I look for when buying a blood pressure watch?
- Look for published validation studies, required calibration frequency, regulatory status in your country, and whether the device measures absolute BP values or only tracks relative trends.