PPG Baroreflex Sensitivity Measurement: Methods, Clinical Value, and Wearable Applications
How PPG measures baroreflex sensitivity using pulse arrival time and inter-beat interval variability. Covers clinical relevance and wearable BRS estimation.
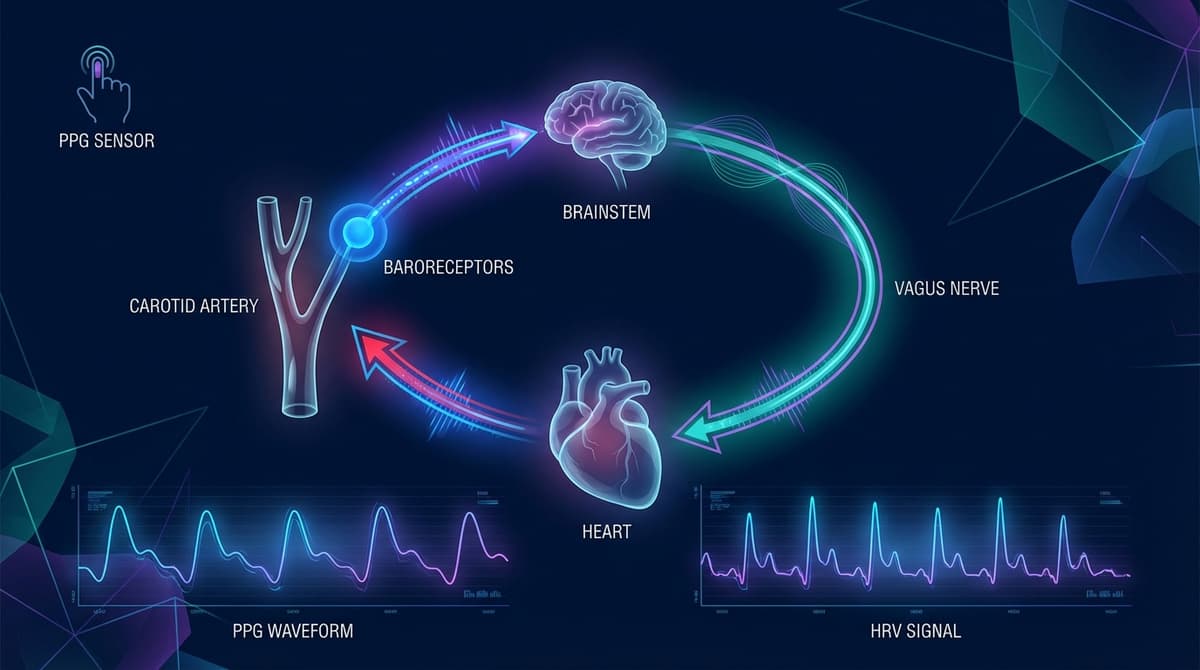
Baroreflex sensitivity (BRS) quantifies how effectively the body adjusts heart rate in response to changes in blood pressure, and photoplethysmography (PPG) can estimate it continuously and non-invasively. By extracting beat-to-beat timing from the PPG waveform, specifically pulse arrival time (PAT) as a surrogate for systolic blood pressure and inter-beat interval (IBI) variability as a measure of cardiac autonomic modulation, wearable devices can approximate BRS without a blood pressure cuff or ECG. This makes PPG-based BRS estimation a practical tool for long-term autonomic monitoring in clinical and home settings. Reduced BRS is a well-established predictor of adverse outcomes after myocardial infarction, in heart failure, and in diabetic autonomic neuropathy, so bringing continuous measurement to wrist-worn devices has real clinical value.
The Baroreflex: A Primer
The arterial baroreflex is one of the body's most important short-term blood pressure regulation mechanisms. Stretch-sensitive receptors (baroreceptors) located in the carotid sinus and aortic arch detect changes in arterial wall distension caused by blood pressure fluctuations. When blood pressure rises, baroreceptor firing increases, which triggers a vagal (parasympathetic) response that slows heart rate and reduces peripheral vascular resistance. When blood pressure drops, baroreceptor firing decreases, and sympathetic activation increases heart rate and constricts blood vessels.
This feedback loop operates on a beat-to-beat timescale. It is fast, reflexive, and continuous. The gain of this loop, how many milliseconds of R-R interval change result from each mmHg change in systolic blood pressure, is what we call baroreflex sensitivity.
A healthy BRS in a young adult is typically 10 to 30 ms/mmHg. Values decline with age. They also decline with cardiovascular disease, diabetes, and conditions that damage autonomic nerve fibers. BRS below 3 ms/mmHg is associated with significantly increased cardiovascular mortality.
Traditional Methods for Measuring BRS
Pharmacological Methods
The Oxford method, introduced by Smyth et al. in 1969, remains the gold standard. It involves injecting phenylephrine (a vasoconstrictor) intravenously to raise blood pressure by 15 to 40 mmHg, then measuring the reflex slowing of heart rate. BRS is calculated as the slope of the regression line between systolic blood pressure and R-R interval over sequential beats. The method is reliable and well-validated, but it requires IV access, continuous invasive or finger-cuff blood pressure monitoring, and clinical supervision.
Nitroprusside can be used to lower blood pressure and assess the sympathetic limb of the baroreflex, though this is less commonly performed in routine testing.
Mechanical Methods
The neck chamber technique applies positive or negative pressure to the neck to mechanically stimulate or unload the carotid baroreceptors. Negative pressure simulates a blood pressure increase (stretching the carotid sinus wall), while positive pressure simulates a drop. This allows selective assessment of carotid baroreflex function without systemic drug administration. It is primarily a research tool.
The Valsalva Maneuver
The Valsalva maneuver (forced expiration against a closed glottis at 40 mmHg for 15 seconds) produces a characteristic four-phase blood pressure response. During strain release (phase II late and phase IV), the overshoot in blood pressure triggers a baroreflex-mediated bradycardia. The Valsalva ratio (longest R-R interval after release divided by shortest R-R interval during strain) provides a crude index of baroreflex function. For more on autonomic maneuvers, see our guide to PPG autonomic function testing.
Spontaneous Sequence Method
The spontaneous sequence method, developed by Bertinieri et al. (1985), avoids any external perturbation. It identifies naturally occurring sequences of three or more consecutive beats where systolic blood pressure and R-R interval both increase (or both decrease) progressively. The slope of each qualifying sequence gives a BRS estimate. This method requires continuous blood pressure and ECG recording but no pharmacological intervention. It forms the conceptual basis for PPG-based approaches.
Transfer Function (Spectral) Method
Rather than looking at sequences in the time domain, the transfer function method calculates the gain between blood pressure and R-R interval oscillations in the frequency domain, typically in the low-frequency band (0.04 to 0.15 Hz) where baroreflex-mediated oscillations are concentrated. This approach uses cross-spectral analysis and provides both gain and phase information.
How PPG Enables Non-Invasive BRS Estimation
Traditional BRS methods all require beat-to-beat blood pressure measurement, which historically meant an arterial catheter or, at minimum, a finger-cuff device like Finapres. PPG changes this by providing two surrogate signals that, when combined, approximate the baroreflex relationship.
Pulse Arrival Time as a Blood Pressure Surrogate
Pulse arrival time (PAT) is the interval from the ECG R-wave (or, in pure PPG systems, from one reference point) to the arrival of the pulse wave at a peripheral site. PAT has an inverse relationship with blood pressure: when blood pressure rises, arterial stiffness increases, pulse wave velocity increases, and the pulse arrives sooner. When blood pressure falls, PAT lengthens.
In PPG-only systems without ECG, pulse transit time (PTT) can be estimated using dual-site PPG (for example, ear and finger) or by using features within the PPG waveform itself. The time from the foot of the waveform to specific fiducial points encodes information about wave propagation speed. For accuracy considerations, see our analysis of PPG inter-beat interval measurement.
Inter-Beat Interval from PPG
The peak-to-peak or foot-to-foot interval of consecutive PPG pulses provides an estimate of R-R interval. While PPG-derived IBI has lower temporal precision than ECG-derived R-R intervals (due to waveform morphology changes and motion artifacts), modern algorithms achieve agreement within 5 to 15 ms under resting conditions. This is sufficient for BRS estimation, which relies on trends over sequences of beats rather than single-beat precision.
PPG-Based Spontaneous Sequence BRS
The most common PPG approach to BRS adapts the spontaneous sequence method. The algorithm works as follows:
- Extract beat-to-beat IBI from the PPG signal (surrogate for R-R interval).
- Extract beat-to-beat PAT or a related pulse timing metric (surrogate for systolic blood pressure).
- Identify sequences of three or more consecutive beats where PAT and IBI move in the same direction (both shortening or both lengthening).
- Calculate the slope of the IBI vs. PAT regression for each qualifying sequence.
- Average across qualifying sequences to produce a BRS estimate.
Because PAT is inversely related to blood pressure, the expected baroreflex relationship is: as PAT decreases (blood pressure rises), IBI should increase (heart rate slows). The slope of this relationship, in milliseconds of IBI change per millisecond of PAT change, can be calibrated against traditional BRS units (ms/mmHg) if a PAT-to-BP transfer function is known.
Spectral Approaches with PPG
Frequency-domain BRS estimation is also feasible with PPG. The transfer function gain between PAT variability and IBI variability in the low-frequency band provides a spectral BRS estimate. This approach is more robust to noise in individual sequences because it uses the entire recording rather than discrete qualifying sequences. Pinheiro et al. (2016) demonstrated that PPG-derived spectral BRS correlated with invasive BRS measurements (r = 0.78) in a cohort of post-MI patients (DOI: 10.1088/0967-3334/37/8/1214).
Validation and Accuracy of PPG-Based BRS
Several studies have compared PPG-derived BRS against gold-standard methods. The results are encouraging, though not without limitations.
Schäfer and Vagedes (2013) conducted a systematic comparison of non-invasive BRS assessment techniques, including PPG-based methods, and found that spontaneous methods (both sequence and spectral) showed moderate to strong correlation with pharmacological BRS (r = 0.60 to 0.85) under resting conditions (DOI: 10.1016/j.ijcard.2012.09.119). The agreement tends to be strongest in populations with normal to mildly reduced BRS and weakest at the extremes.
Key sources of error in PPG-based BRS include:
- Motion artifacts: Even small movements introduce noise into PAT and IBI estimates, which can create false qualifying sequences or obscure real ones. This is the primary barrier to ambulatory PPG-BRS monitoring.
- Respiratory confounding: Respiration modulates both blood pressure and heart rate independently of the baroreflex. Without respiratory gating, PPG-BRS estimates may conflate respiratory sinus arrhythmia with baroreflex-mediated variability.
- PAT vs. true blood pressure: PAT reflects both pulse transit time (vascular) and pre-ejection period (cardiac). Changes in pre-ejection period (due to contractility shifts) can alter PAT without any change in blood pressure, introducing error.
- Low temporal resolution: PPG sampling rates of 25 to 50 Hz (common in wearables) limit PAT precision to 20 to 40 ms, which reduces sensitivity to small BRS changes.
Despite these limitations, PPG-based BRS provides clinically useful stratification. It reliably distinguishes between high, normal, and low BRS categories even if absolute values carry more uncertainty than invasive methods.
Clinical Applications
Post-Myocardial Infarction Risk Stratification
Reduced BRS after myocardial infarction is one of the strongest predictors of sudden cardiac death and all-cause mortality. The ATRAMI study established that BRS below 3 ms/mmHg, combined with reduced heart rate variability, identifies a high-risk subgroup with a relative risk of cardiac mortality exceeding 8. Continuous PPG-based BRS monitoring during cardiac rehabilitation could flag patients whose autonomic recovery is stalling, enabling earlier intervention.
Heart Failure Monitoring
In heart failure, sympathetic overactivation and reduced vagal tone progressively impair baroreflex function. BRS declines as heart failure worsens, making it a potential marker of disease progression. Wearable PPG devices worn during sleep (when motion artifacts are minimal) could track BRS trends over weeks and months, providing an early warning of decompensation before symptoms become overt. For related cardiac monitoring approaches, see our overview of PPG and the baroreceptor reflex.
Diabetic Autonomic Neuropathy
Cardiac autonomic neuropathy (CAN) is a common and underdiagnosed complication of diabetes. It damages the vagal fibers that mediate the baroreflex, leading to reduced BRS, resting tachycardia, and exercise intolerance. Screening for CAN currently requires dedicated autonomic testing in a clinic. PPG-based BRS, combined with HRV analysis from wearables, could enable routine screening at scale, catching CAN in its early, potentially reversible stages.
Hypertension and Aging
BRS decreases with age and is further reduced in hypertension. Tracking BRS alongside blood pressure in treated hypertensive patients could help clinicians assess whether antihypertensive therapy is restoring autonomic balance, not just lowering numbers. Some antihypertensives (ACE inhibitors, beta-blockers) improve BRS, while others have neutral or negative effects. Longitudinal PPG-BRS data could inform treatment selection.
Sleep and Circadian Autonomic Profiling
BRS normally increases during sleep, reflecting enhanced vagal tone. Blunted nocturnal BRS augmentation is seen in obstructive sleep apnea, heart failure, and autonomic neuropathy. Wrist-worn PPG devices that record overnight are ideally positioned to capture this circadian BRS pattern. The sleep context also minimizes the motion artifact problem that plagues daytime ambulatory recordings.
Wearable Implementation Considerations
Building BRS estimation into a consumer wearable requires addressing several practical challenges.
Sampling rate matters. Most consumer wearables sample PPG at 25 Hz. For BRS estimation, higher rates (50 to 100 Hz) improve PAT precision and reduce quantization noise in the slope calculation. Some devices offer burst-mode high-frequency sampling during rest periods, which is a reasonable compromise between power consumption and signal quality.
Signal quality gating is essential. Not every segment of PPG data is suitable for BRS estimation. An effective implementation must detect and discard segments corrupted by motion, poor sensor contact, or arrhythmia. Quality metrics such as perfusion index, signal-to-noise ratio, and waveform template matching can automate this gating.
Context annotation improves interpretation. BRS varies with posture, activity, stress, and sleep stage. A raw BRS number without context is hard to interpret. Wearables that combine PPG-BRS with accelerometer-derived activity classification, body position detection, and time-of-day metadata can present BRS in meaningful context: resting supine BRS, standing BRS, nocturnal BRS, and post-exercise BRS recovery.
Population-level calibration is needed. Because PPG-BRS uses PAT as a blood pressure surrogate, the absolute BRS values (in ms/mmHg) require calibration. This can be done at the population level using age, sex, and blood pressure reference data, or at the individual level if the user performs an occasional cuff measurement to anchor the PAT-BP relationship.
Frequently Asked Questions
What is baroreflex sensitivity and why does it matter?
Baroreflex sensitivity measures how effectively the heart rate changes in response to blood pressure fluctuations. It is expressed in milliseconds of R-R interval change per mmHg of systolic blood pressure change. High BRS indicates a responsive autonomic nervous system. Low BRS reflects impaired autonomic regulation and is associated with increased risk of cardiac arrhythmias, sudden death after heart attack, and progression of heart failure. It is one of the most powerful autonomic markers for cardiovascular risk stratification.
Can a smartwatch really measure baroreflex sensitivity?
A smartwatch cannot measure BRS with the same precision as a clinical setup involving continuous blood pressure monitoring and ECG. However, it can estimate BRS using surrogate signals extracted from PPG: pulse arrival time (which tracks blood pressure changes) and inter-beat intervals (which track heart rate changes). Under controlled conditions, particularly during rest or sleep, these estimates correlate meaningfully with gold-standard BRS and can reliably categorize individuals into low, normal, and high BRS groups.
How is PPG-based BRS different from HRV analysis?
HRV analysis examines the variability of heart rate on its own. BRS specifically measures the coupling between blood pressure changes and heart rate responses. A person can have preserved HRV but impaired BRS if the heart rate variability is driven by respiration or other non-baroreflex mechanisms. Conversely, reduced HRV often accompanies reduced BRS, but the two metrics provide complementary information. PPG-based BRS adds the blood pressure surrogate dimension that pure HRV analysis lacks.
What is a normal baroreflex sensitivity value?
Normal BRS ranges from approximately 10 to 30 ms/mmHg in healthy young adults. It declines with age: typical values for a 60-year-old might be 5 to 10 ms/mmHg. Values below 6 ms/mmHg are considered reduced, and values below 3 ms/mmHg are associated with significantly elevated cardiovascular risk. These thresholds were established primarily using the phenylephrine method and the spontaneous sequence technique. PPG-derived surrogate values may require device-specific reference ranges.
Does posture affect PPG-based BRS measurement?
Yes. BRS is significantly influenced by posture. Standing reduces BRS compared to supine rest because the sympathetic nervous system is more active during upright posture. PPG-based BRS measurements should always be interpreted in the context of body position. For the most reliable trending, measurements should be compared within the same postural condition, for example, supine BRS measured during sleep on consecutive nights.
What clinical conditions cause reduced baroreflex sensitivity?
Multiple conditions impair BRS. These include myocardial infarction (especially in the acute phase and the first year post-MI), chronic heart failure, essential hypertension, diabetes mellitus (through cardiac autonomic neuropathy), obstructive sleep apnea, chronic kidney disease, and aging itself. Certain medications like atropine block the vagal limb of the baroreflex. Conversely, regular aerobic exercise, weight loss, and some medications (ACE inhibitors, statins) can improve BRS.
How often should BRS be measured with a wearable device?
For clinical trending, nightly measurements during sleep offer the best combination of signal quality and consistency. Sleep recordings minimize motion artifacts and provide a standardized autonomic state. Weekly or monthly trends in nocturnal BRS are more clinically meaningful than single measurements. In research settings, multiple 5-minute resting recordings per day can capture circadian BRS variation. The optimal frequency depends on the clinical question: post-MI monitoring might warrant daily tracking, while general wellness screening could rely on weekly summaries.