Photoplethysmography Sensor: Types, Design, and Selection Guide
Everything you need to know about photoplethysmography sensors: LED wavelengths, photodetector types, analog front-ends, form factors, and how to choose the right sensor for your application.
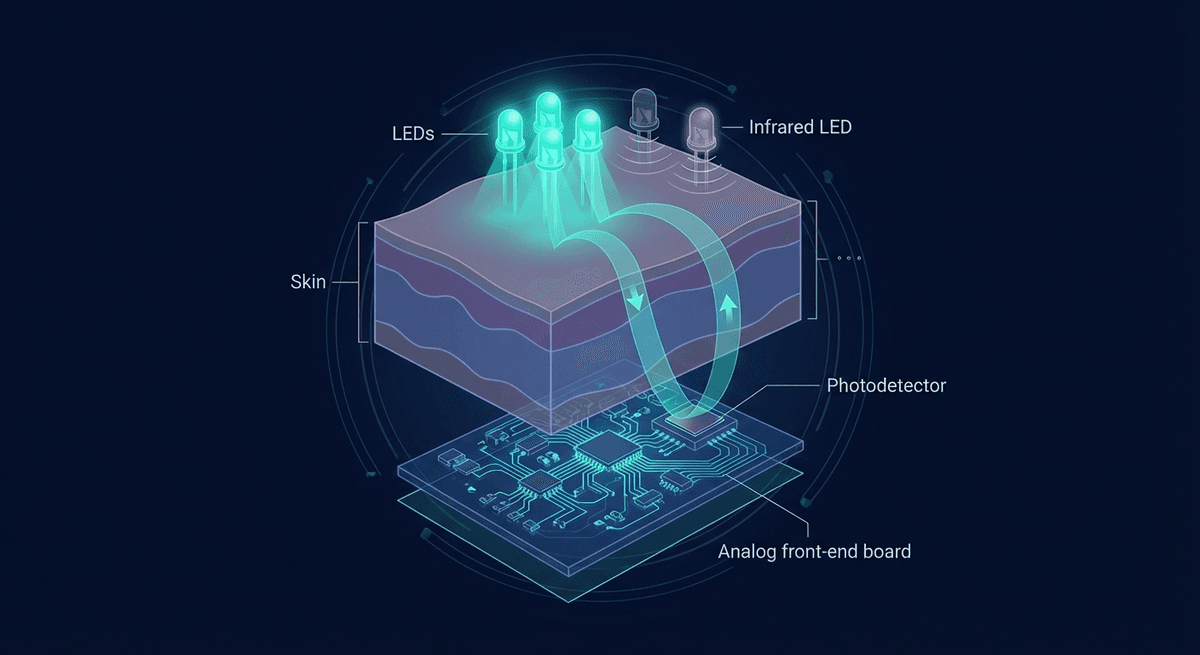
A photoplethysmography sensor is an optoelectronic assembly that measures changes in blood volume in tissue by detecting how much light is absorbed or reflected. At its core, it is deceptively simple: an LED, a photodetector, and an analog front-end to convert the tiny photocurrent into a usable signal. The engineering challenge lies in getting clean cardiac-synchronous signal out of a noisy, motion-prone environment at microwatt power budgets.
This guide covers the full hardware stack of a PPG sensor — from LED selection and photodetector design through analog front-end circuits, form factors, and how to pick the right sensor for different applications.
The Basic PPG Sensor Architecture
Every PPG sensor has three functional layers:
- Illumination: One or more LEDs that inject light into the tissue
- Detection: A photodetector that captures returned (reflected or transmitted) light
- Signal conditioning: Amplification, filtering, and ADC to convert photocurrent to digital data
In reflectance mode (wrist wearables), the LED and detector are co-located on the same surface. In transmission mode (fingertip pulse oximeters), they are on opposite sides of the tissue.
LED Selection: Wavelength Matters
The choice of LED wavelength is the most consequential design decision in PPG sensor engineering. Different wavelengths penetrate tissue to different depths and are absorbed by different chromophores.
Green (515–570 nm)
The workhorse wavelength for consumer wrist-based heart rate monitoring. Hemoglobin (both oxy and deoxy forms) absorbs green light strongly, producing a large pulsatile signal. The high extinction coefficient means a strong AC/DC ratio — more of the detected signal variation is cardiac-synchronous.
Tradeoffs: Green light has shallow tissue penetration (~1–2 mm), so it primarily samples subepidermal capillaries and small arterioles. It is more affected by melanin than longer wavelengths, contributing to reduced signal amplitude in darker skin tones. It is also more sensitive to motion artifact from surface tissue movement.
Common green LED wavelengths: 520 nm (peak hemoglobin absorption in the green band), 530 nm, 550 nm.
Red (620–700 nm)
Standard clinical pulse oximetry uses 660 nm. Oxyhemoglobin (HbO₂) and deoxyhemoglobin (Hb) have very different absorption at 660 nm — HbO₂ absorbs less, Hb absorbs much more. This differential absorption is what makes SpO₂ calculation possible when combined with an NIR wavelength.
Red light penetrates deeper than green (~3–5 mm), sampling larger arterioles and venules. Less affected by melanin. Weaker pulsatile signal than green at equivalent power, but better suited for transmission-mode measurements and SpO₂.
Near-Infrared (800–950 nm)
Used paired with red for SpO₂ (standard: 940 nm). At ~805 nm (the isobestic point), HbO₂ and Hb absorb equally — useful as a reference. At 940 nm, HbO₂ absorbs more than Hb, opposite to the 660 nm pattern.
NIR penetrates the deepest (~5–10 mm), sampling larger vessels. Low melanin absorption makes it well-suited for devices used across skin tones. The weaker hemoglobin extinction coefficient at NIR wavelengths means the pulsatile signal is smaller relative to green, but this is offset by the deeper penetration and better SNR at melanin-rich sites.
Multi-Wavelength Designs
Modern medical-grade sensors use three or four wavelengths (e.g., 470 nm blue + 530 nm green + 660 nm red + 880 nm NIR). Benefits:
- Wavelength-switching can identify the cleanest signal channel in real time
- Multiple wavelengths enable SpO₂ plus heart rate plus vascular metrics simultaneously
- Redundancy improves resilience to motion artifact
- Research into six-wavelength PPG has shown potential for estimating hemoglobin concentration
Devices like the Masimo W1 and Samsung Galaxy Watch Ultra use multi-wavelength arrays. Research platforms like the PLUX biosignals system offer full spectral flexibility.
Photodetector Design
Photodiode vs. Phototransistor
Photodiodes dominate PPG applications due to:
- Faster response (photodiodes respond to MHz signals; cardiac PPG only needs <100 Hz, but photodiodes also handle LED modulation frequencies of 100 kHz+)
- Lower noise at small signals
- Better temperature stability
- More predictable linearity
Phototransistors have higher gain but also higher noise and slower response — not ideal for precision PPG.
Active Area and Geometry
Larger photodetector active area captures more light, improving signal-to-noise ratio. But larger detectors also pick up more ambient light noise. Optimal detector size is a function of the optical geometry and shielding design.
Many sensors use multiple small detectors in an array rather than one large detector. This allows spatial filtering — detectors far from the LED see less motion artifact from surface tissue and more signal from deeper blood vessels.
Optical Shielding
Ambient light rejection is critical. Design approaches:
- Black housing between LED and detector to prevent direct optical crosstalk
- Optical bandpass filters over the detector, passing only LED wavelengths
- Light seals at skin contact to minimize ambient light ingress
- Electronic ambient cancellation: sample with LED off, subtract from LED-on measurement
Most consumer wearable sensors achieve ambient rejection of 40–60 dB.
Analog Front-End (AFE)
The AFE converts the tiny photodiode current into a clean digital signal suitable for DSP processing.
Transimpedance Amplifier (TIA)
The photodiode generates a current proportional to light intensity — typically in the range of 10 nA to 10 µA for a well-designed reflectance PPG sensor. A transimpedance amplifier (TIA) converts this current to voltage with a gain of 10 kΩ to 1 MΩ.
The TIA design determines the noise floor. Key metric: input-referred noise current, typically targeted below 1 pA/√Hz in medical-grade AFEs.
Programmable Gain and LED Drive
The pulsatile AC component represents only 0.5–5% of the total detected light (DC component dominates). High-quality AFEs use:
- Cancellation of the DC photocurrent before amplification (allows higher gain on AC component without saturation)
- Programmable LED drive current (typically 0 to 100 mA in 100 µA steps)
- Auto-gain control that adjusts LED current and TIA gain to keep the signal in optimal range across skin tones and sensor placements
LED Modulation
Pulsing the LED at high frequency (typically 1–10 kHz, far above the cardiac signal) and synchronously detecting (lock-in amplification) separates LED-generated signal from broadband ambient light. The AFE integrates multiple LED pulses per sample, improving SNR proportionally to the square root of the integration count.
Key AFE Chips
| Chip | Manufacturer | Key Features |
|---|---|---|
| AFE4404 | Texas Instruments | 3 LED channels, 16-bit ADC, I2C/SPI |
| MAX86141 | Maxim/Analog Devices | 2 LED channels, ultra-low noise, 19-bit ADC |
| MAX30101 | Maxim | Integrated LED + detector, SpO₂ + HR |
| AFE4900 | Texas Instruments | ECG + PPG on one chip, biosensing |
| SFH7060 | ams-OSRAM | Integrated LED array + detector module |
Form Factors and Applications
Fingertip Clip (Transmission Mode)
Medical standard for pulse oximetry and clinical heart rate. Best signal quality. Restricts movement — not suitable for continuous ambulatory use.
Wrist Band
Consumer standard for 24/7 monitoring. Reflectance mode. Susceptible to motion artifact. Green LEDs dominant for heart rate; multi-wavelength for SpO₂. Sensor placement matters (inside of wrist preferred, ~1–2 cm proximal to wrist crease, not over the radius bone).
Ring
Emerging form factor. Oura Ring, Samsung Galaxy Ring, RingConn. Finger-based reflectance or transmission, better signal-to-noise than wrist, lower motion artifact in sleep, but restricted fingers limit use in some activities.
Earlobe / Ear Canal
Earable PPG. Concha and ear canal sensors offer stable placement, low motion artifact, and access to the auricular artery. Bose SleepBuds, Jabra, and research earbud platforms have explored this. Signal quality is excellent for stable resting monitoring.
Chest Patch
Some medical patches (Philips BioTel Heart, iRhythm Zio) combine ECG and PPG in a single adhesive patch. The chest is not ideal for PPG (less pulsatile signal than finger or ear) but provides multi-modal data.
Forehead Clip
Hospital use for patients with poor peripheral perfusion. Forehead has rich blood supply from the temporal and supraorbital arteries. Less susceptible to peripheral vasoconstriction than finger sensors.
Choosing the Right PPG Sensor
Consider these factors:
| Factor | Choice Impact |
|---|---|
| Application (SpO₂ vs. HR only) | Single wavelength (HR) vs. dual wavelength (SpO₂) |
| Regulatory requirements | Consumer: no clearance needed. Clinical: FDA/CE required |
| Skin tone diversity | Multi-wavelength or NIR-dominant preferred |
| Power budget | LED current dominates power; optimize modulation duty cycle |
| Motion tolerance | Multi-wavelength + accelerometer fusion essential for exercise |
| Signal quality requirements | Medical grade: high-res AFE, careful optical design; consumer: integrated modules OK |
Frequently Asked Questions
What wavelength does Apple Watch use for PPG? Apple Watch uses multiple LEDs including green (~520 nm) for heart rate and red + near-infrared (approximately 660 nm and 940 nm) for blood oxygen (SpO₂). The specific chip design is proprietary, but Apple has described using an array of green, red, and infrared LEDs in their patents.
Why do PPG sensors use green light for heart rate? Green light (~520–550 nm) is strongly absorbed by hemoglobin, producing a large pulsatile signal that is easy to detect. For reflectance measurement at the wrist, it offers the best signal-to-noise ratio in most individuals under low-motion conditions.
What is the best PPG sensor for a wearable research project? For prototyping: MAXM86161 (Maxim) or AFE4404 (TI) are common choices in academia. For integrated modules: MAX30102 or SFH7050 are widely available breakout boards. For production: work with a sensor vendor (ams-OSRAM, OSRAM, TI, Analog Devices) for application-specific optimization.
Can a PPG sensor measure blood pressure directly? No PPG sensor measures blood pressure directly — there is no optical principle that yields absolute pressure. Cuffless blood pressure estimation uses PPG-derived features (pulse transit time, waveform morphology) combined with calibration against a reference BP measurement. It is indirect and requires individual calibration for accuracy.
How does skin tone affect PPG sensor performance? Melanin absorbs light across all visible wavelengths, especially shorter wavelengths. Green LEDs are most affected — darker skin tones produce smaller pulsatile signals with green LEDs, requiring higher LED drive current and higher-gain amplification. NIR wavelengths (>800 nm) are much less affected by melanin, making them more equitable across skin tones.
References
-
Tamura, T., Maeda, Y., Sekine, M., & Yoshida, M. (2014). Wearable photoplethysmographic sensors—past and present. Electronics, 3(2), 282–302. https://doi.org/10.3390/electronics3020282
-
Mendelson, Y., & Ochs, B. D. (1988). Noninvasive pulse oximetry utilizing skin reflectance photoplethysmography. IEEE Transactions on Biomedical Engineering, 35(10), 798–805. https://doi.org/10.1109/10.7286
-
Webster, J. G. (Ed.). (1997). Design of Pulse Oximeters. Taylor & Francis. ISBN 0750303875.
-
Bent, B., Goldstein, B. A., Kibbe, W. A., & Dunn, J. P. (2020). Investigating sources of inaccuracy in wearable optical heart rate sensors. npj Digital Medicine, 3, 18. https://doi.org/10.1038/s41746-020-0226-6