PPG Biomarkers in Type 2 Diabetes: Vascular Stiffness, Microcirculation, and CV Risk
How PPG reveals cardiovascular risk in type 2 diabetes: arterial stiffness indices, microvascular dysfunction detection, autonomic neuropathy assessment, and wearable monitoring applications.
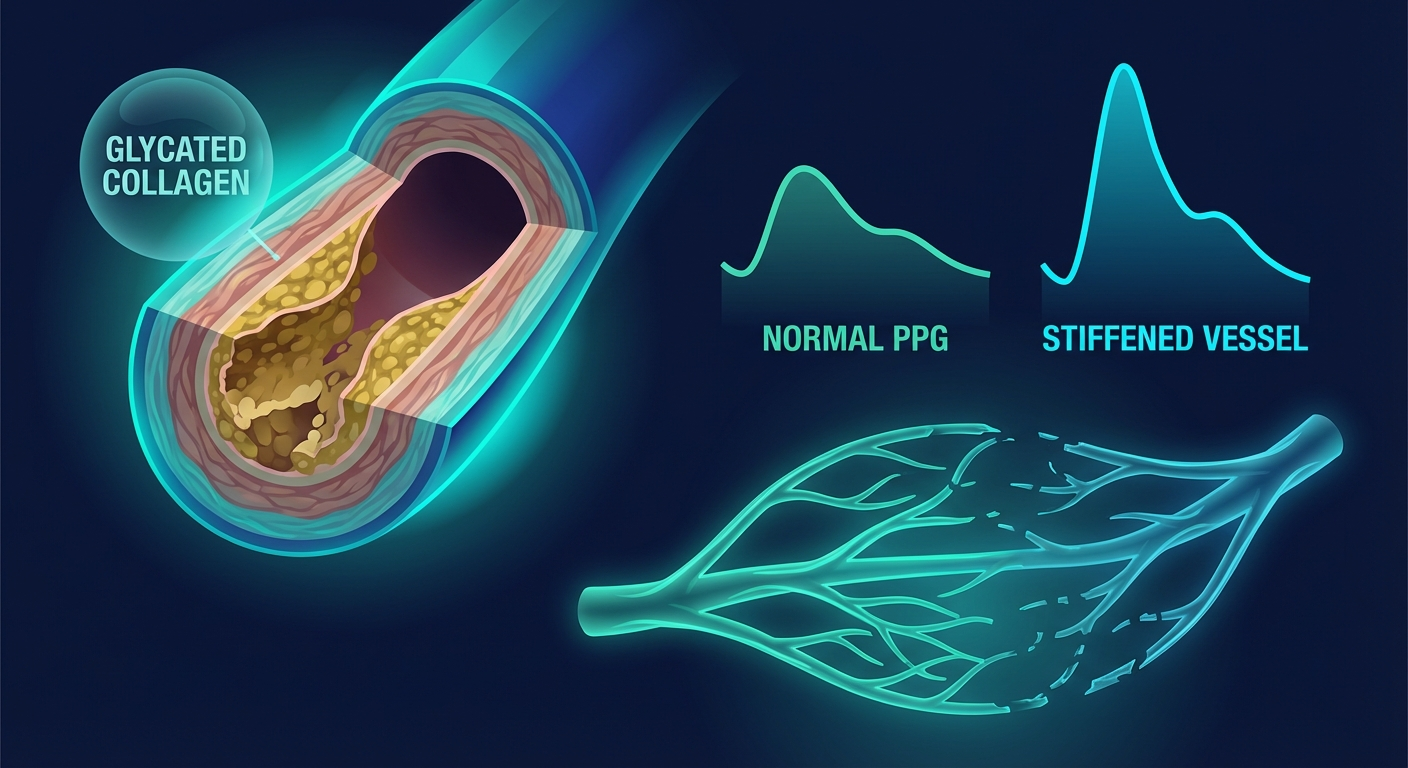
Type 2 diabetes mellitus carries a two-to-four times higher cardiovascular risk than the general population, driven largely by accelerated vascular aging, autonomic neuropathy, and microvascular dysfunction. PPG biomarkers can non-invasively characterize several of these pathological processes, making them potentially useful for risk stratification and monitoring in people with diabetes.
The relationship between diabetes and PPG is bidirectional: diabetes damages the vessels and nerves that determine PPG signal characteristics, and those changes are detectable before clinical complications manifest. This makes PPG a candidate for early detection and longitudinal monitoring.
Arterial Stiffness in Diabetes: What PPG Reveals
Hyperglycemia accelerates vascular aging through several mechanisms: glycation of structural proteins (collagen, elastin) in vessel walls, oxidative stress from advanced glycation end products (AGEs), and chronic inflammation. The result is a stiffer arterial tree that manifests in the PPG signal.
Augmentation Index (AIx)
The augmentation index, derived from PPG waveform analysis, quantifies the proportion of pulse pressure attributable to wave reflection from the periphery. In a stiff aorta, reflected waves return faster (shorter round-trip time) and arrive during systole rather than diastole, augmenting the systolic peak and increasing cardiac afterload.
AIx is calculated from the second derivative (SDPPG) or direct second-derivative analysis of the PPG waveform. Multiple studies have established that:
- AIx is elevated in people with type 2 diabetes compared to age-matched controls
- AIx elevation correlates with HbA1c and duration of diabetes
- Each 10% elevation in AIx corresponds to increased major adverse cardiovascular event risk
A study by Cecelja and Chowienczyk (2012, doi:10.1161/CIRCRESAHA.111.302000) reviewed the evidence linking aortic stiffness (as measured by PWV and AIx) to cardiovascular outcomes in diabetes and confirmed that aortic stiffness is an independent predictor of cardiovascular events in diabetic populations beyond traditional risk factors.
Pulse Wave Velocity (PWV)
Pulse wave velocity is the gold-standard measure of arterial stiffness. PPG-based PTT provides an accessible proxy. In type 2 diabetes, PWV is consistently elevated: studies show PWV 1–3 m/s higher in type 2 diabetics compared to non-diabetic controls of the same age.
The clinical significance: each 1 m/s increase in carotid-femoral PWV is associated with a 14% increase in cardiovascular events in the general population. In diabetes, where baseline PWV is already elevated, this risk escalation is compounded.
Wrist-worn PPG devices cannot measure PWV directly (which requires two measurement sites), but they can track PTT trends as a proxy for stiffness changes over time, particularly in response to lifestyle or pharmacological interventions.
Microvascular Dysfunction
The microvasculature (capillaries and arterioles) is a primary target of diabetic damage. Microvascular dysfunction in diabetes precedes and contributes to retinopathy, nephropathy, and neuropathy, and also contributes to macrovascular cardiovascular risk through endothelial dysfunction.
PPG can reveal microvascular changes through several features:
Reduced perfusion index: Diabetic microvascular disease reduces capillary density and increases arteriolar tone, reducing the pulsatile component of the PPG signal. Lower resting perfusion index in people with diabetes is documented in multiple studies.
Reduced dicrotic notch prominence: The dicrotic notch in the PPG waveform reflects the aortic valve closure and the interaction of incident and reflected waves. In people with microvascular dysfunction, the dicrotic notch becomes less distinct and may disappear, reflecting altered vascular compliance and wave reflection patterns.
Altered waveform slopes: The systolic upstroke slope (related to cardiac output and compliance) and diastolic decay slope (related to peripheral resistance and compliance) are both altered in diabetic arteriopathy.
A 2020 study by Elgendi et al. (doi:10.3389/fphys.2020.00392) reviewed PPG waveform indices in diabetes and confirmed that morphological features including the augmentation index, inflection point area ratio, and notch/trough ratios are all significantly altered in people with type 2 diabetes compared to controls.
Diabetic Autonomic Neuropathy (DAN)
Diabetic autonomic neuropathy affects approximately 20–30% of people with type 2 diabetes and is one of the most significant but underdiagnosed complications. DAN damages the sympathetic and parasympathetic nerves innervating the cardiovascular system, producing:
- Resting tachycardia (due to parasympathetic denervation)
- Reduced HRV
- Orthostatic hypotension
- Inability to mount appropriate heart rate responses to exercise
PPG-derived HRV is a practical screening tool for cardiac autonomic neuropathy (CAN). The cardiovascular autonomic reflex tests (CARTs) standardized by Ewing et al. include measures directly assessable from PPG-derived pulse rate variability:
- Deep breathing test: HRV during deep breathing at 6 breaths/minute. Expiration:inspiration ratio < 1.17 at age 20–40 is abnormal.
- Valsalva ratio: Heart rate acceleration during straining compared to deceleration after release.
- 30:15 ratio: Heart rate acceleration at 15th beat after standing compared to deceleration at 30th beat.
These tests, originally requiring ECG, can be adapted for PPG with moderately reduced precision. A study by Röttger et al. (2022) found that pulse-based CART testing showed 84% agreement with ECG-based testing for CAN classification.
The clinical importance of detecting CAN is high: people with CAN have a 5-year mortality approximately 2–3 times higher than diabetic patients without CAN, primarily from cardiac arrhythmias and silent myocardial infarction.
Glycemic Variability and PPG
Blood glucose fluctuations have acute effects on vascular function that are detectable in PPG. Post-meal glucose spikes cause transient endothelial dysfunction (oxidative stress, reduced nitric oxide production) that manifests as reduced PPG amplitude and increased peripheral vascular resistance.
Research by Ceriello et al. has demonstrated that glycemic variability, not just average glucose, independently predicts cardiovascular outcomes. PPG waveform monitoring during continuous glucose monitoring could, in principle, capture the vascular consequences of glycemic excursions in real time.
Some research groups have explored whether PPG signals can be used to estimate glucose levels non-invasively (see the related article on PPG glucose monitoring). While direct glucose estimation from PPG remains unproven for clinical use, the correlation between glycemic state and PPG vascular indices opens an interesting window into metabolic-cardiovascular coupling.
Wearable Monitoring Applications for Diabetic Patients
Given the pathological relationships described, several PPG monitoring applications are relevant for people with type 2 diabetes:
Daily HRV monitoring for autonomic neuropathy progression: Annual CAN screening with standardized autonomic reflex tests, supplemented by daily wearable HRV monitoring to track longitudinal trends. Declining HRV trend is an early warning sign of progressing autonomic neuropathy.
Resting heart rate monitoring: Elevated resting heart rate (reflecting parasympathetic denervation) is a marker of CAN. A consistent rise in resting HR over months, without other explanation, warrants clinical evaluation.
Activity response monitoring: People with severe CAN have blunted heart rate responses to exercise. Wearable monitoring during exercise can characterize the heart rate response profile and flag abnormally blunted responses.
Nocturnal pattern monitoring: Non-dipping blood pressure and non-dipping heart rate (no overnight reduction) are common in diabetes with autonomic neuropathy and predict cardiovascular events. PPG-based overnight heart rate monitoring can detect non-dipping patterns.
Post-intervention assessment: After lifestyle intervention (weight loss, exercise), medications (metformin, GLP-1 agonists, SGLT2 inhibitors), or intensive glucose control, serial PPG vascular indices provide objective evidence of cardiovascular risk factor improvement.
Limitations
PPG vascular assessments are sensitive but not specific for diabetes. Aging, hypertension, smoking, and renal disease all produce similar changes to arterial stiffness and autonomic function. Distinguishing diabetic from non-diabetic vascular changes requires clinical context.
Additionally, most of the PPG vascular assessment work has been done with research-grade clinical devices rather than consumer wearables. The accuracy of consumer devices for detecting subtle waveform morphology changes in diabetes is less well established.
Internal Links
- For arterial stiffness measurement: PPG Aortic Stiffness Biomarker
- For the second derivative analysis: PPG Second Derivative SDPPG
- For autonomic testing methods: PPG Autonomic Function Testing
FAQ
Can PPG detect diabetic complications early? PPG can detect vascular changes associated with diabetes, including elevated arterial stiffness, reduced perfusion index, and reduced HRV, that correlate with diabetic complication risk. These changes appear before clinical symptoms, making PPG a potential tool for early risk detection. However, PPG alone cannot diagnose diabetes or specific complications.
What is diabetic autonomic neuropathy and how is PPG used to detect it? Diabetic autonomic neuropathy (DAN) damages the nerves controlling heart rate and blood pressure. It manifests as reduced HRV, resting tachycardia, and impaired heart rate responses to standing and breathing. PPG-derived pulse rate variability can assess these responses through cardiovascular autonomic reflex tests.
How does high blood sugar damage blood vessels, and can PPG measure this? Hyperglycemia causes glycation of vessel wall proteins, oxidative stress, and inflammation, all of which stiffen and thicken blood vessels. PPG reflects this through reduced dicrotic notch prominence, elevated augmentation index, and altered waveform slopes, though consumer devices may not detect subtle early changes reliably.
Is wearable heart rate variability monitoring useful for people with diabetes? Yes. Daily HRV monitoring provides a practical window into autonomic function. Consistently low HRV in a diabetic patient may indicate cardiac autonomic neuropathy, a serious but often underdiagnosed complication. Longitudinal HRV trends can also monitor the cardiovascular response to diabetes management improvements.
Can smartwatch PPG replace clinical vascular testing for diabetic patients? No. Clinical vascular assessments (pulse wave velocity, ankle-brachial index, autonomic reflex testing) provide more precise and validated measurements for clinical decision-making. Smartwatch PPG is appropriate for longitudinal trend monitoring and general wellness tracking, not clinical diagnostic testing.
What PPG changes are most characteristic of type 2 diabetes? The most consistent PPG changes in type 2 diabetes are: elevated augmentation index, reduced dicrotic notch prominence, shortened pulse transit time (reflecting increased stiffness), reduced resting HRV, and reduced perfusion index. These changes are more pronounced with longer diabetes duration and poorer glycemic control.